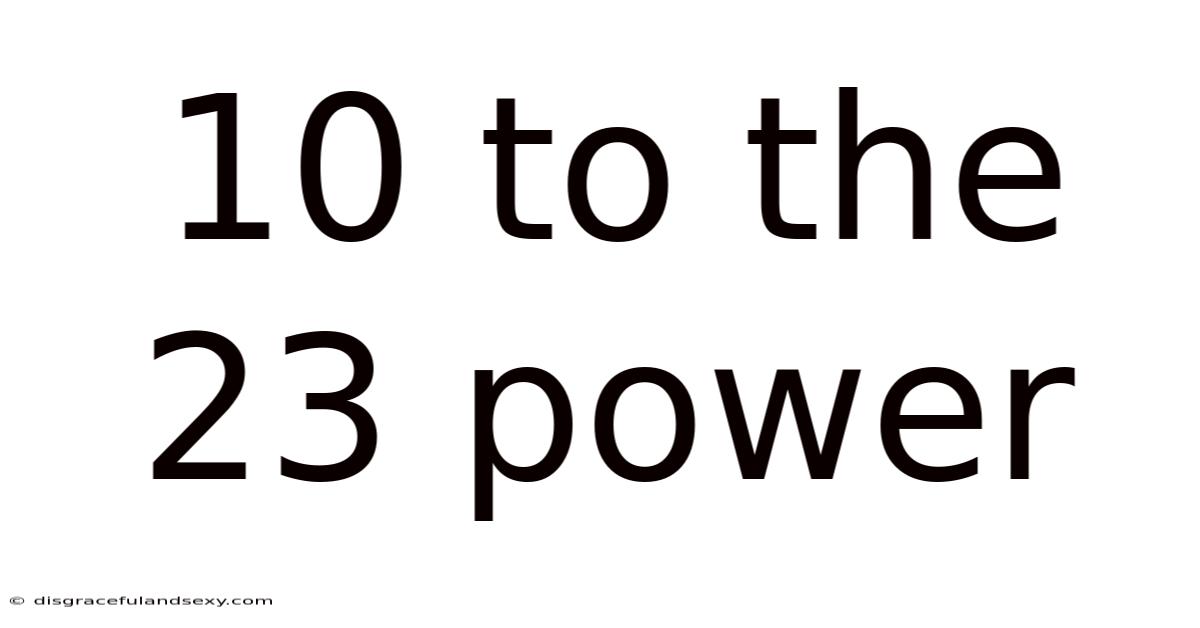Delving into the Immense: Understanding 10 to the Power of 23
Have you ever considered the sheer scale of numbers beyond our everyday comprehension? Worth adding: imagine a number so vast it dwarfs even the largest figures we encounter in daily life. This article digs into the concept of 10<sup>23</sup>, exploring its magnitude, its relevance in various scientific fields, and the implications of understanding such exceptionally large numbers. Now, we'll uncover how this number relates to everyday objects, the universe, and the fundamental building blocks of matter. Understanding 10<sup>23</sup> isn't just about numbers; it's about grasping the immense scale of the universe and the remarkable precision of scientific measurement Practical, not theoretical..
Understanding Exponential Notation
Before we dive into the specifics of 10<sup>23</sup>, let's establish a firm understanding of exponential notation. Think about it: this is a shorthand way of representing extremely large (or very small) numbers. The expression 10<sup>23</sup> signifies 10 multiplied by itself 23 times. This is a far more convenient way to write the number than writing out all the zeros: 10,000,000,000,000,000,000,000,000 It's one of those things that adds up..
This type of notation is crucial in scientific fields where dealing with extraordinarily large or small quantities is commonplace. From the number of atoms in a grain of sand to the distance to distant galaxies, exponential notation provides a manageable and efficient system for representing these vast differences in scale.
Worth pausing on this one.
10<sup>23</sup> in the Context of Avogadro's Number
One of the most prominent applications of 10<sup>23</sup> lies within the realm of chemistry, specifically in the concept of Avogadro's number. Avogadro's number is approximately 6.Because of that, 022 x 10<sup>23</sup>. This number represents the number of constituent particles (atoms, molecules, ions, or other particles) in one mole of a substance. A mole is a fundamental unit in chemistry, representing a specific quantity of matter.
Imagine trying to count the number of atoms in even a tiny amount of a substance. It would be an impossible task without the concept of the mole and Avogadro's number. This number provides a practical and standardized way to work with quantities of matter at the atomic and molecular level. It bridges the gap between the macroscopic world we experience and the microscopic world of atoms and molecules Not complicated — just consistent..
Some disagree here. Fair enough.
Relating 10<sup>23</sup> to Everyday Objects
While 10<sup>23</sup> might seem abstract, we can relate it to everyday objects to grasp its magnitude. Let's consider grains of sand. A single grain of sand is incredibly small, but even a handful contains a vast number of them. While the exact number varies depending on the size of the grain and the size of the hand, it's safe to say that a handful of sand would contain far fewer particles than 10<sup>23</sup> Worth keeping that in mind. That's the whole idea..
To truly appreciate the size of 10<sup>23</sup>, consider scaling up. Now, even this colossal number would still fall significantly short of 10<sup>23</sup>. Practically speaking, imagine a beach, encompassing countless grains of sand. Now imagine thousands of beaches, each filled with as many grains of sand as the first. This exercise illustrates the immense scale we are dealing with Easy to understand, harder to ignore..
This is where a lot of people lose the thread Worth keeping that in mind..
Applications of 10<sup>23</sup> in Physics and Astronomy
The number 10<sup>23</sup> isn't limited to chemistry; it extends its reach into physics and astronomy as well. In astrophysics, this scale helps us comprehend the vastness of the universe. The number of stars in a galaxy, for instance, is often in the order of billions, or 10<sup>9</sup> to 10<sup>12</sup>. Although vastly large, these numbers are still relatively small compared to 10<sup>23</sup> Small thing, real impact..
Some disagree here. Fair enough.
Adding to this, 10<sup>23</sup> can be used to represent the number of particles in certain astronomical phenomena. These cosmic structures contain an incredibly large number of particles, a number which could well exceed 10<sup>23</sup> in many cases. Consider the scale of nebulae, giant clouds of gas and dust where stars are born. The density of such cosmic structures, even in relatively sparse regions, is incomprehensible without the context of these large numbers.
People argue about this. Here's where I land on it That's the part that actually makes a difference..
The Importance of Scientific Notation in Handling Such Numbers
Working with numbers like 10<sup>23</sup> highlights the critical importance of scientific notation. Which means without it, calculations and comparisons would become incredibly cumbersome and error-prone. Which means scientific notation allows scientists to perform calculations involving extremely large or small numbers efficiently and accurately. It simplifies complex equations and facilitates a clear understanding of relative magnitudes Easy to understand, harder to ignore..
The use of scientific notation isn't merely a matter of convenience; it's a necessity. And it forms the backbone of many scientific calculations and is essential for accurate data representation and analysis. The precision and efficiency offered by scientific notation are invaluable tools in various scientific disciplines Worth keeping that in mind..
Exploring the Limits of Human Comprehension
One of the challenges in dealing with numbers like 10<sup>23</sup> is their sheer unfathomable scale. Our brains are simply not wired to intuitively grasp quantities of this magnitude. We struggle to visualize or relate to such enormous numbers because they far exceed our everyday experience.
On the flip side, the pursuit of understanding these immense numbers is crucial for advancing our knowledge of the universe. Practically speaking, by employing mathematical tools, models, and analogies, scientists strive to make sense of these colossal figures and their implications. This effort is not just about understanding the numbers themselves; it's about understanding the processes and phenomena they represent.
Beyond the Number: The Implications of Scale
The significance of 10<sup>23</sup> extends beyond its numerical value. It represents a scale of magnitude that forces us to reconsider our perception of the universe. It highlights the vastness of space, the intricacies of matter, and the power of scientific methods to unravel the complexities of the natural world.
Understanding numbers like 10<sup>23</sup> challenges our intuition and expands our understanding of the universe's scale. It allows us to appreciate the immense complexity of even seemingly simple things like a grain of sand, and to comprehend the scale of processes in chemistry, physics, and astrophysics.
You'll probably want to bookmark this section.
Frequently Asked Questions (FAQ)
- Q: What is the full written-out form of 10<sup>23</sup>?
A: Writing out 10<sup>23</sup> in full would be 10,000,000,000,000,000,000,000,000,000. As you can see, this is impractical and prone to errors. Scientific notation is far more efficient and accurate.
- Q: Can we visualize 10<sup>23</sup> in any meaningful way?
A: Direct visualization is difficult. Still, analogies like comparing it to the number of grains of sand on a beach (although far less than 10<sup>23</sup>) can help provide a sense of scale, albeit a very rough approximation Simple, but easy to overlook..
- Q: Why is Avogadro's number important?
A: Avogadro's number (approximately 6.022 x 10<sup>23</sup>) provides a bridge between the macroscopic world (grams, moles) and the microscopic world (atoms, molecules). It allows chemists to work with measurable quantities of substances at the atomic and molecular levels.
- Q: Are there numbers larger than 10<sup>23</sup> in scientific contexts?
A: Absolutely! Numbers far exceeding 10<sup>23</sup> are routinely used in fields like cosmology to describe the number of atoms in the observable universe, which is estimated to be around 10<sup>80</sup>.
- Q: How does understanding 10<sup>23</sup> help in everyday life?
A: While not directly applicable to everyday tasks, understanding this scale enhances our appreciation for the vastness of the universe and the precision of scientific measurement. It fosters a greater sense of wonder and encourages critical thinking about scale and magnitude.
Conclusion
Understanding the magnitude of 10<sup>23</sup> is a journey into the realm of the immense. By grasping this scale, we not only enhance our scientific literacy but also develop a deeper appreciation for the complexity and beauty of the natural world. While the number itself may seem abstract, its relevance in various scientific fields is undeniable. From Avogadro's number to the vastness of space, this number represents a scale that challenges our intuition but simultaneously expands our understanding of the universe. The exploration of such large numbers reminds us that our understanding of the universe is a continuous journey of discovery and that even the seemingly incomprehensible can be approached with the right tools and perspective Surprisingly effective..