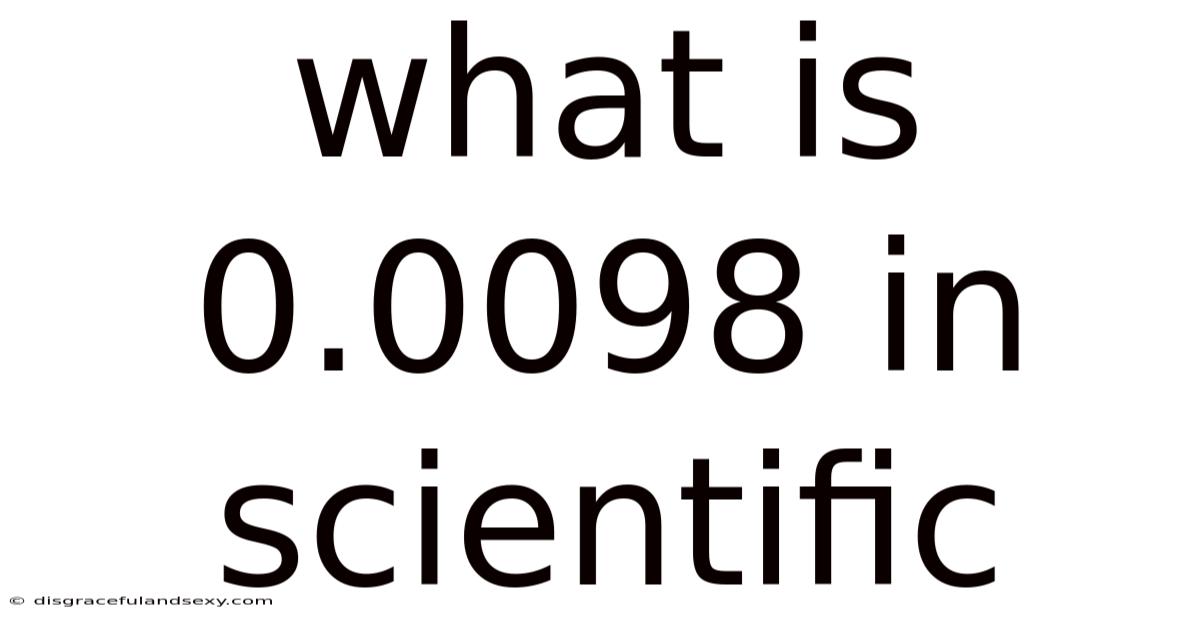What is 0.0098 in Scientific Notation? A complete walkthrough
Understanding scientific notation is crucial for anyone working with very large or very small numbers, common in fields like science, engineering, and computer science. This article will comprehensively explain how to convert the decimal number 0.Day to day, 0098 into scientific notation, covering the underlying principles and providing a deeper understanding of this essential mathematical tool. We'll also address frequently asked questions and explore practical applications.
Not obvious, but once you see it — you'll see it everywhere.
Introduction to Scientific Notation
Scientific notation, also known as standard form, is a way of expressing numbers that are either too large or too small to be conveniently written in decimal form. It's a standardized method that simplifies the representation of these numbers, making them easier to read, write, and manipulate. The general form of a number expressed in scientific notation is:
a x 10<sup>b</sup>
where:
- a is a number between 1 and 10 (but not including 10), also known as the coefficient or significand.
- b is an integer, representing the exponent or power of 10.
Converting 0.0098 to Scientific Notation: A Step-by-Step Guide
Let's break down the process of converting 0.Day to day, 0098 into scientific notation. The key is to manipulate the decimal point to obtain a coefficient (a) between 1 and 10.
Step 1: Identify the Coefficient (a)
We need to move the decimal point in 0.0098 to the right until we get a number between 1 and 10. Moving the decimal point three places to the right gives us 9.8. Which means, our coefficient (a) is 9.8.
Step 2: Determine the Exponent (b)
The exponent (b) represents how many places we moved the decimal point. Since we moved it three places to the right, the exponent is -3. A negative exponent indicates that the original number was less than 1 It's one of those things that adds up. And it works..
Step 3: Write the Number in Scientific Notation
Combining the coefficient (a) and the exponent (b), we express 0.0098 in scientific notation as:
9.8 x 10<sup>-3</sup>
Deeper Dive: Understanding the Exponent
The exponent in scientific notation is crucial for understanding the magnitude of the number. A positive exponent indicates a large number (greater than 1), while a negative exponent indicates a small number (less than 1). The absolute value of the exponent tells you the number of places the decimal point has been moved.
In our example, the exponent of -3 signifies that 0.Plus, 0098 is a small number, three orders of magnitude smaller than 1. Plus, this means it is 1/1000th (one thousandth) of 9. 8 Worth keeping that in mind..
Significance and Applications of Scientific Notation
Scientific notation is indispensable in various scientific and engineering disciplines. Here are a few key applications:
-
Astronomy: Dealing with vast distances and incredibly large numbers (e.g., distances between stars, size of galaxies). To give you an idea, the distance to the nearest star, Proxima Centauri, is approximately 4.243 light-years, which in kilometers is approximately 4.011 x 10<sup>13</sup> km Small thing, real impact..
-
Physics: Handling extremely small quantities like atomic radii or elementary particle masses. The mass of an electron, for instance, is about 9.109 x 10<sup>-31</sup> kg Nothing fancy..
-
Chemistry: Representing the concentration of solutions or the number of molecules in a substance. Avogadro's number, the number of particles in one mole of a substance, is approximately 6.022 x 10<sup>23</sup>.
-
Computer Science: Working with large datasets or representing very small probabilities The details matter here..
-
Engineering: Calculating dimensions of extremely large structures (like bridges or skyscrapers) or very small components (like microchips).
Working with Numbers in Scientific Notation: Addition, Subtraction, Multiplication, and Division
While expressing numbers in scientific notation is beneficial, performing calculations requires understanding how to manipulate them Small thing, real impact..
Addition and Subtraction: To add or subtract numbers in scientific notation, they must have the same exponent. If they don't, adjust one or both numbers to have a matching exponent before performing the calculation. Then, simply add or subtract the coefficients and keep the exponent the same.
Multiplication: To multiply numbers in scientific notation, multiply the coefficients and add the exponents.
Division: To divide numbers in scientific notation, divide the coefficients and subtract the exponents Worth keeping that in mind..
Examples of Calculations with Scientific Notation
Let's consider a few examples to illustrate these operations:
Example 1: Addition
(2.That said, 5 x 10<sup>3</sup>) + (4. That's why 0 x 10<sup>3</sup>) = (2. 5 + 4.0) x 10<sup>3</sup> = 6.
Example 2: Subtraction
(7.1 x 10<sup>-2</sup>) = (7.2 - 3.2 x 10<sup>-2</sup>) - (3.1) x 10<sup>-2</sup> = 4.
Example 3: Multiplication
(3.0 x 10<sup>4</sup>) x (2.So 0 x 2. 0 x 10<sup>2</sup>) = (3.0) x 10<sup>(4+2)</sup> = 6.
Example 4: Division
(8.4 x 10<sup>5</sup>) / (2.Consider this: 1 x 10<sup>2</sup>) = (8. Worth adding: 4 / 2. 1) x 10<sup>(5-2)</sup> = 4 That alone is useful..
Frequently Asked Questions (FAQ)
Q1: Why is scientific notation important?
A1: Scientific notation provides a concise and efficient way to represent extremely large or small numbers, making them easier to work with and understand. It simplifies calculations and improves readability, particularly in scientific and engineering applications Simple as that..
Q2: Can any number be written in scientific notation?
A2: Yes, any number can be expressed in scientific notation. On the flip side, it is most useful and efficient for very large or very small numbers Easy to understand, harder to ignore..
Q3: What if the coefficient is not between 1 and 10?
A3: If your coefficient is not between 1 and 10, you need to adjust the decimal point and the exponent accordingly to bring the coefficient within the required range Simple, but easy to overlook..
Q4: How do I convert a number from scientific notation to decimal form?
A4: To convert a number from scientific notation to decimal form, simply move the decimal point in the coefficient to the right (for a positive exponent) or to the left (for a negative exponent) by the number of places indicated by the absolute value of the exponent.
Not obvious, but once you see it — you'll see it everywhere.
Q5: Are there any limitations to scientific notation?
A5: While very effective, scientific notation doesn't directly display the precision of a number in the same way as decimal notation. The number of significant figures needs to be carefully managed to maintain accuracy That alone is useful..
Conclusion
Scientific notation is a powerful tool for representing and manipulating very large and very small numbers. Understanding its principles, including the manipulation of coefficients and exponents, is essential for success in various scientific, engineering, and computational fields. Think about it: this guide provided a comprehensive overview, from the basic conversion of 0. Worth adding: 0098 to a deeper exploration of its applications and calculations. Practically speaking, mastering this system significantly enhances your ability to handle and comprehend numerical data across diverse disciplines. Remember to practice conversions and calculations regularly to build proficiency and confidence in using scientific notation effectively.